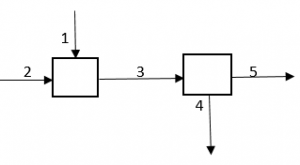
Question
a.
1
b.
3
c.
6
d.
8
Posted under Basic Chemical Engineering
Interact with the Community - Share Your Thoughts
Uncertain About the Answer? Seek Clarification Here.
Understand the Explanation? Include it Here.
Q. How many independent equations are possible if each stream contains mixture of A, B and C?
Similar Questions
Explore Relevant Multiple Choice Questions (MCQs)
Q. 1 has pure A and 2 has pure B and 3, 4 and 5 has mixture of A and B, how many independent equations are possible?
View solution
Q. How many independent equations are possible, if two components are involved in each of streams?
View solution
Q. How many independent equations are possible, if only one component exists in each stream?
View solution
Q. Answer the following question for the diagram.
What is the value of F?
View solution
Q. Answer the following question for the diagram given below.
What is the value of W?
View solution
Q. What is the value of C?
View solution
Q. Answer the following questions for the given diagram.
What is the value of B?
View solution
Q. What is the value of H/P in the following process, if 2P + H = 16?
View solution
Q. What is the value of F*P in the following process?
View solution
Q. What is the value of P in the following process?
View solution
Q. What is the value of P in the following process ?
View solution
Q. What is the value of P in the following process?
View solution
Q. Methane is at the rate 50 moles is supplied to a reactor with air at the rate 1000 moles, the reactor leaves O₂, N₂, CO₂ and H₂O, what is the percentage of H₂O in products?
View solution
Q. Methane is at the rate 50 moles is supplied to a reactor with air at the rate 1000 moles, the reactor leaves O₂, N₂, CO₂ and H₂O, what is the percentage of N₂ in products?
View solution
Q. Methane is at the rate 50 moles is supplied to a reactor with air at the rate 1000 moles, the reactor leaves O₂, N₂, CO₂ and H₂O, what is the percentage of CO₂ in products?
View solution
Q. Methane is at the rate 50 moles is supplied to a reactor with air at the rate 1000 moles, the reactor leaves O₂, N₂, CO₂ and H₂O, what is the percentage of O₂ in products?
View solution
Q. Methane is at the rate 50 moles is supplied to a reactor with air at the rate 1000 moles, the reactor leaves O₂, N₂, CO₂ and H₂O, what is the rate of products?
View solution
Q. octane is burnt with 40% excess O₂, what is the percentage of CO₂ in products?
View solution
Q. Propane is burnt with 20% excess O₂, what is the percentage of CO₂ in products?
View solution
Q. Pentane is burnt with 100% of excess air, what is the percentage of H₂O in the products?
View solution
Recommended Subjects
Are you eager to expand your knowledge beyond Basic Chemical Engineering? We've handpicked a range of related categories that you might find intriguing.
Click on the categories below to discover a wealth of MCQs and enrich your understanding of various subjects. Happy exploring!